Type of Posting: General Announcement
Posting Date: 04-Jun–2007
Official Date: 01-May-2013
The Nomenclature Expert Committee has voted to adopt revisions to General Chapter <1121> Nomenclature. These revisions will appear in USP 31, available in November 2007. USP is making these upcoming revisions available to the public in advance in order to allow companies additional time to bring pipeline products that have not yet been approved by FDA into compliance with the policy.
Background
The USP Nomenclature Expert Committee establishes the official titles for dosage form monographs in the USP–NF. These titles generally become the recognized established names (generic names) for the dosage forms.
For many years, USP's Nomenclature Expert Committee used a policy called the Salt Nomenclature Policy in establishing monograph titles for drug products that use a salt form as the active pharmaceutical ingredient. In Pharmacopeial Forum 32(4) [July–Aug. 2006], USP proposed to include the text of that policy in General Chapter <1121> Nomenclature in order to broaden understanding of the policy among all parties who may be affected by it, including regulators, manufacturers, and practitioners. As with all proposed revisions of the USP–NF, the Expert Committee welcomed comments from the public, including manufacturers and health care practitioners.
Following the public comment period, the Expert Committee reviewed the comments received and decided to change the policy as shown below. They also voted to make the new policy official on May 1, 2013, in order to allow manufacturers to bring the common or usual names of their pipeline products into conformance with the new policy, so that the nonproprietary name submitted to FDA at the time of product approval will be consistent with the established name that will be assigned by USP.
New Policy
The Committee adopted the following new policy, to replace the Salt Nomenclature Policy:
MONOGRAPH NAMING POLICY FOR SALT DRUG SUBSTANCES IN DRUG PRODUCTS AND COMPOUNDED PREPARATIONS
The titles of USP monographs for drug products and compounded preparations formulated with a salt of an acid or base use the name of the active moiety, as defined below. The strength of the product or preparation also is expressed in terms of the active moiety.
An active moiety is the molecule or ion, excluding those appended portions of the molecule that cause the drug to be an ester, salt (including a salt with hydrogen or coordination bonds), or other noncovalent derivative (such as a complex, chelate, or clathrate) of the molecule, responsible for the physiological or pharmacological action of the drug substance, without regard to the actual charged state of the molecule in-vivo.
For example, the active moiety of a hydrochloride salt of a base will be the free base and not the protonated form of the base. The active moiety of a metal acid salt will be the free acid.
-
Example: Chelocardin Hydrochloride active moiety is Chelocardin
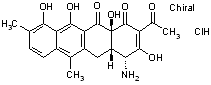

-
Example: Alendronate Sodium active moiety is Alendronic Acid
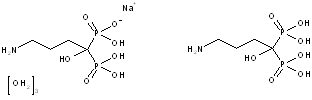
This Policy is followed by USP in naming drug products and compounded preparations that are newly recognized in the USP. Revising existing monographs to conform to this Policy is not intended, except where the USP Council of Experts determines that, for reasons such as safety, a nomenclature change is warranted.
Related Issues
Labeling. The labeling clearly states the specific salt form of the active moiety that is present in the product/preparation, as this information may be useful to practitioners and patients. The names and strengths of both the active moiety and specific salt form (where applicable) are provided in the labeling.
Exceptions. In those rare cases in which the use of the specific salt form of the active moiety in the title provides vital information from a clinical perspective, an exception to this Policy may be considered. In such cases, where the monograph title contains the specific salt form of the active moiety, the strength of the product or preparation also is expressed in terms of the specific salt form.
In addition, the Committee adopted the following implementation scheme for the new policy:
IMPLEMENTATION PROCESS FOR MONOGRAPH NAMING POLICY
-
For title requests coming to the NOM EC on 1 May 2013 and thereafter
Apply the Monograph Naming Policy for Salt Drug Substances in Drug Products and Compounded Preparations (Policy) as provided in General Chapter <1121> Nomenclature in USP 31 and NF 26. This policy will become official 1 May 2013. -
For title requests coming to the NOM EC before 1 May 2013, the official date of the Policy
Use the interim established names1 with respect to active moiety/salt title/strength terminology in USP monograph titles for all dosage form monographs that are proposed or implemented between 1 March 2007 and 1 May 2013 (the official date of the Policy), except in cases where safety issues are involved or where the relevant manufacturers in the U.S. market agree to a title that conforms to the Policy. In this period leading to implementation of the Policy, however, USP strongly encourages industry to adopt titles conforming to the Policy in order to increase consistency and decrease practitioners' confusion.Cancel, upon request, any USP-proposed dosage form monograph title that (1) was proposed before 1 March 2007, but is not yet official, and (2) differs from the interim established name for that dosage form, and propose a new monograph title that conforms to the interim established name with respect to active moiety/salt title/strength terminology, except in cases where safety issues are involved. The NOM EC will review such safety issue cases on an individual basis.
As an example, if the USP Nomenclature Expert Committee (NOM EC) receives a request for the title "[Drug] Sodium Tablets," the interim established name, during this 1 March 2007 to 1 May 2013 time period, the NOM EC will approve "[Drug] Sodium Tablets" as the monograph title, even if the product strength is expressed in terms of the free acid. This Policy also directs the NOM EC, upon request, to use the interim established name for titles that were previously proposed but have not yet become official. In this case, if the proposed monograph title was "[Drug] Acid Tablets," that title would become "[Drug] Sodium Tablets."
1. See Novartis Pharmaceuticals Corp. v. Leavitt, 435 F.3d 344 (D.C. Cir. 2006).
USP will work with the Food and Drug Administration (FDA) to ensure that products receiving FDA approval use names that comply with this new Policy.
For further information, please contact Dr. W. Larry Paul, USP Department of Healthcare Quality and Safety, Standards Division, at 1+301-816-8331, fax 1+301-816-8373, or wlp@usp.org
